Immune checkpoint inhibitors for the treatment of unresectable stage III NSCLC
Author:
Prof. Dr. med. Miklos Pless
Senior Consultant Medizinische Onkologie und Hämatologie, Kantonsspital Winterthur
Sie sind bereits registriert?
Loggen Sie sich mit Ihrem Universimed-Benutzerkonto ein:
Sie sind noch nicht registriert?
Registrieren Sie sich jetzt kostenlos auf universimed.com und erhalten Sie Zugang zu allen Artikeln, bewerten Sie Inhalte und speichern Sie interessante Beiträge in Ihrem persönlichen Bereich
zum späteren Lesen. Ihre Registrierung ist für alle Unversimed-Portale gültig. (inkl. allgemeineplus.at & med-Diplom.at)
Immune checkpoint inhibitors (ICIs) have fundamentally changed treatment algorithms in lung cancer, irrespective of histological subtype and disease stage. Stage IIIB/C non-small cell lung cancer (NSCLC) is generally defined as locally advanced disease without hematogenous metastases (M0). It is considered unresectable but can potentially be cured with a combination of concurrent radiotherapy and chemotherapy (cCRT). In this article, we discuss the role of ICIs in unresectable stage III NSCLC, including their efficacy, the associated challenges, open questions, and future directions. Stage III NSCLC with actionable driver mutations will not be addressed.
Keypoints
-
ICI have dramatically improved outcomes in unresectable stage III NSCLC.
-
Concurrent ICI with definitive RT is not recommended.
-
The role of induction ICI/CT before cCRT is currently being investigated.
-
Many de-escalation strategies offer options for specific patient populations.
Historical background
One of the first attempts to cure locally advanced NSCLC was described in the paper by the Veterans Administration Group.1 In this randomized trial, 554 patients were assigned either to local RT with 40–50Gy or no local treatment. Keep in mind that CT scans for staging were not yet available, and most centers used orthovoltage radiotherapy. The patients included were allowed to have a performance status (PS) of up to 3, and all types of histology were eligible, including small cell lung cancer (SCLC). Nevertheless, a small but significant survival benefit with the use of RT (median 14 days) was observed. This trial laid the foundation of radiotherapy (RT) in unresectable, locally advanced NSCLC.
The following five decades were characterized by improvements in many areas, e.g. trial methodology, such as the separation of SCLC as a distinct entity, or the restriction of patient inclusion to PS 0/1. Advances of diagnostic tools included the introduction of CT and PET-CT scans. Important innovations in radiotherapy were also made, such as the use of megavoltage techniques, 3D/4D RT, and more recently intensity-modulated RT (IMRT) and volumetric-modulated arc therapy (VMAT). Finally, the addition of cisplatin-containing chemotherapy (CT) to RT was established. After a long transatlantic dispute on whether the two modalities should be delivered sequentially or concomitantly, the West Japan Lung Cancer Group could show that concurrent chemoradiotherapy (cCRT) with 56Gy was superior to the sequential application and resulted in a 5-year overall survival (OS) rate of 16%.2 What followed was a sobering 15-year period, during which it proved extremely difficult to further improve the results mentioned above. Both induction3 and consolidation CT4 failed, as did increasing the RT dose from 60 to 72Gy,5 or using more efficacious drugs, such as pemetrexed, in combination with cisplatin.6 Clearly, a novel therapeutic concept was needed.
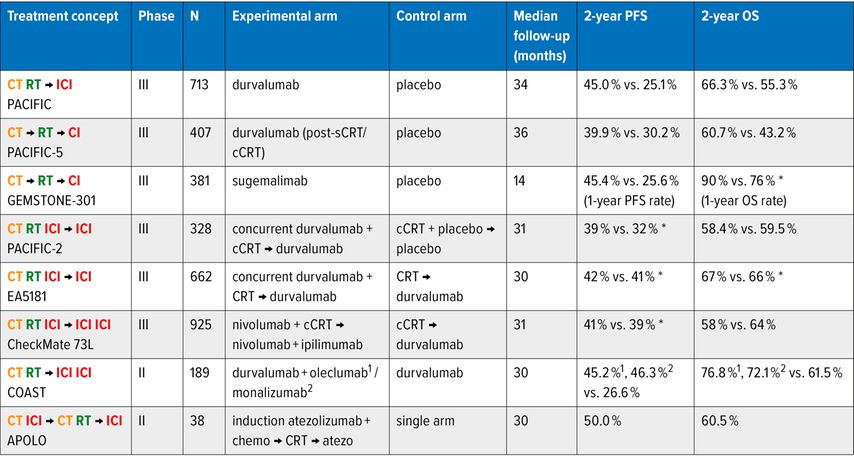
Tab. 1: Unresectable stage III NSCLC: immunotherapy strategies and landmark outcomes (2-year PFS/OS where available); *estimated from Kaplan-Meier curve
The era of ICI
This novel concept proved to be the addition of ICI to cCRT. In the pivotal PACIFIC study, patients whose tumor had not progressed after standard platinum-containing cCRT with 60–66Gy, were randomized to receive either durvalumab for one year or placebo. The results were impressive: mature median and 5-year progression-free survival (PFS) improved from 5.6 to 16.9 months and from 19% to 33% , respectively. OS improved as well from 29.1 to 47.5 months and from 33% to 43% , respectively. This benefit was seen across most subgroups, possibly with the exception of PD-L1-negative tumors.7 Therefore, the PACIFIC regimen is the undisputed new standard of care. Attempts to further improve the regimen were initiated rapidly. The following questions were addressed in clinical trials:
Is sequential CRT followed by consolidation ICI also effective?
Two randomized phase II trials investigated this question: PACIFIC-5 with durvalumab8 and GEMSTONE 301 using sugemalimab, a Chinese PD-L1 inhibitor.9 Both studies included a placebo control group and were positive for their primary endpoint of improved PFS. Both trials also showed a trend towards improved OS, but the results are still immature and appear less impressive than those of the original PACIFIC trial. The current recommendation is therefore to use the cCRT regimen, if feasible.
Is there a role for concurrent CRT/ICI followed by consolidation ICI?
Three independent phase III trials examined this question: PACIFIC-2,10 EA518111 and CheckMate 73L.12 The first two trials used durvalumab as ICI during CRT and for consolidation, while CheckMate 73L used nivolumab during CRT and ipilimumab/nivolumab for consolidation, thus also exploring an intensified consolidation strategy. Remarkably, despite the positive results of the original PACIFIC trial, the control arm in PACIFIC-2 received placebo. In contrast, EA5181 and CheckMate 73L used durvalumab, which is the correct comparator. Surprisingly, none of these trials showed a survival benefit for the experimental arm with cCRT plus ICI, with 3-year OS results similar to PACIFIC. Several explanations have been considered:
-
Increased toxicity, especially pneumonitis, when radiotherapy is administered together with ICI.13
-
A negative immunomodulatory effect of irradiating the tumor-draining lymph nodes.14
-
Direct killing of activated T-effector cells at the irradiated tumor site.15
-
In the PACIFIC trial randomization was performed after cCRT and only in patients with at least stable disease, which meant that the 27% of the patients who did not complete cCRT or had tumor progression were excluded from the analysis. In contrast, in PACIFIC-2, EA5181 and CheckMate 73L, randomization was performed at the beginning of cCRT, so that these presumably frailer patients or more resistant tumors were included.
The currently available evidence indicates that ICIs should not be administered concurrently with cCRT.
Intensification of consolidation therapy
This strategy was tested in the CheckMate 73L trial, by adding ipilimumab to nivolumab, but the results were disappointing.12 In the phase II COAST study, two novel substances were added to durvalumab consolidation. The first was oleclumab, an anti-CD73 monoclonal antibody that prevents the immunosuppressive conversion of AMP to adenosine by CD73. The second was monalizumab, an antibody directed against NKG2A, which is an inhibitor of T and NK cells. Both combinations improved PFS compared to durvalumab alone, but showed no clear benefit in OS.16 The phase III PACIFIC-9 trial will hopefully give a definitive answer regarding the efficacy of these two antibodies. Currently, intensification of consolidation should only be performed within clinical trials.
Induction with chemoimmunotherapy
Similarly to stage II/IIIA NSCLC, where neoadjuvant chemoimmunotherapy is established, induction has been investigated in several phase II trials for unresectable stage III NSCLC.
The APOLO study was a single-arm phase II trial using an induction therapy with atezolizumab plus carboplatin/paclitaxel for three cycles before cCRT, followed by one year of atezolizumab maintenance.17 The overall response rate (ORR) to induction was 47%, with only 13% of patients experiencing progressive disease (PD). The 1- and 2-year PFS rates were 68% and 50% respectively; OS rates at one and two years were 87% and 61%.
The InTRist study was a randomized phase II trial in which patients in the experimental arm were given toripalimab, a PD-1 inhibitor, together with platinum-based chemotherapy for two cycles, followed by cCRT and one year of toripalimab consolidation.18 Interestingly, the control arm also included an induction treatment with chemotherapy alone. After a median follow-up of only 14.7 months, the 1-year PFS rate was 86% vs. 55% in the control arm, and ORR was 78% vs. 40%. These promising results justify further evaluation in phase III trials.
The BRIDGE trial (NCT04765709) is one of them. Unfortunately, after the induction with IO/CT, the patients will be given RT plus IO, but no chemotherapy.
Induction chemoimmunotherapy without consolidation
The CA209-7AL feasibility trial investigated two cycles of induction chemoimmunotherapy with cisplatin/docetaxel and nivolumab, followed by cCRT. The experimental arm was just observed thereafter, whereas the control arm received one year of nivolumab consolidation.19 There was a clear difference in PFS at 12 months in favor of the consolidation arm (73% vs. 61%). These results suggest that the addition of ICI consolidation improved the results notably, and that consolidation therapy should not be omitted outside clinical trials.
Induction and consolidation with ICI alone
In the single-arm phase II trial AFT-16, patients were treated with four cycles of induction ICI (atezolizumab) followed by cCRT and one year of atezolizumab consolidation.20 The outcomes in patients who completed cCRT were quite promising, but they must be interpreted with caution: of the 62 initially included patients, only 76% completed the induction therapy and only 71% completed cCRT. Thus, further data are needed for this approach.
ICI induction, RT alone, followed by ICI consolidation
This is a promising option for patients with a high PD-L1 expression who cannot tolerate chemotherapy. It was tested in the small phase II trial SPRINT.21 Only patients with tumors with a PD-L1 expression >50% were eligible. They received three cycles of pembrolizumab induction followed by PET-based dose-painted RT and 36 weeks of adjuvant pembrolizumab. The ORR to ICI induction was 48%; the 1-year PFS rate was 76%, with a median PFS of 26 months. One- and two-year OS rates were 92% and 76%, respectively. This was a very small trial, but the results were maintained with a median follow-up of >30 months and warrant further investigation.
RT followed by ICI consolidation
In patients unfit to tolerate CT, the concept of RT followed by one year of ICI consolidation with durvalumab, was tested in the DUART study.22 One-year PFS and OS rates were 40% and 65%, respectively, but the median follow-up at 11 months was still quite short. These results are interesting, especially because patients with PD-L1-negative or -low tumors were included as well, but confirmation in larger cohorts is required.
CT/ICI followed by ICI consolidation without RT
For patients with tumors with a high PD-L1 tumor proportion score (TPS), ≥50%, the possibility of omitting RT was tested in the single-arm phase II trial Evolution.23 In this study, patients received four cycles of pembrolizumab together with standard chemotherapy followed by two years of pembrolizumab maintenance therapy. After a median follow-up of 33 months, the 2-year PFS and OS rates were 67% and 81%, respectively. These results are intriguing, but with only 21 patients, this trial is too small to change practice. At present, it is unclear whether cure is possible without radiotherapy or whether it is a palliative treatment with extended PFS and OS.
Conclusion
Just as in the metastatic setting, ICIs have brought enormous changes in the treatment of early-stage NSCLC. In the setting of operable NSCLC, neoadjuvant ICIs are now standard of care, and as recent studies show, ICIs can successfully shift the limits of surgically treated patients to borderline resecetable stage III NSCLC.24 For locally advanced NSCLC, the PACIFIC trial has defined a new standard of care. Multiple trials are trying to improve the PACIFIC results. So far, it is safe to say that ICIs should not be given concomitantly with RT, but induction ICI and various de-escalation strategies might be promising options.
Literatur:
1 Roswit B et al.: The survival of patients with inoperable lung cancer: a large-scale randomized study of radiation therapy versus placebo. Radiology 1968; 90(4): 688-97 2 Furuse K et al.: Phase III study of concurrent versus sequential thoracic radiotherapy in combination with mitomycin, vindesine, and cisplatin in unresectable stage III non-small-cell lung cancer. J Clin Oncol 1999; 17(9): 2692-9 3 Vokes EE et al.: Induction chemotherapy followed by chemoradiotherapy compared with chemoradiotherapy alone for regionally advanced unresectable stage III Non-small-cell lung cancer: Cancer and Leukemia Group B. J Clin Oncol 2007; 25(13): 1698-704 4 Hanna N et al.: Phase III study of cisplatin, etoposide, and concurrent chest radiation with or without consolidation docetaxel in patients with inoperable stage III non-small-cell lung cancer: the Hoosier Oncology Group and U.S. Oncology. JClin Oncol 2008; 26(35): 5755-60 5 Bradley JD et al.: Standard-dose versus high-dose conformal radiotherapy with concurrent and consolidation carboplatin plus paclitaxel with or without cetuximab for patients with stage IIIA or IIIB non-small-cell lung cancer (RTOG 0617): a randomised, two-by-two factorial phase 3 study. Lancet Oncol 2015; 16(2): 187-99 6 Senan S et al.: PROCLAIM: randomized phase III trial of pemetrexed-cisplatin or etoposide-cisplatin plus thoracic radiation therapy followed by consolidation chemotherapy in locally advanced nonsquamous non-small-cell lung cancer. J Clin Oncol 2016; 34(9): 953-62 7 Spigel DR et al.: Five-year survival outcomes from the PACIFIC trial: durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. J Clin Oncol 2022; 40(12): 1301-11 8 Wu YL et al.: PACIFIC-5: aphase III clinical trial of consolidation durvalumab in patients with unresectable stage III NSCLC and no progression after concurrent or sequential chemoradiotherapy. JHematol Oncol 2025; 18(1): 111 9 Zhou Q et al.: Sugemalimab versus placebo after concurrent or sequential chemoradiotherapy in patients with locally advanced, unresectable, stage III non-small-cell lung cancer in China (GEMSTONE-301): interim results of a randomised, double-blind, multicentre, phase 3 trial. Lancet Oncol 2022; 23(2): 209-19 10 Bradley JD et al.: Simultaneous durvalumab and platinum-based chemoradiotherapy in unresectable stage III non-small cell lung cancer: the phase III PACIFIC-2 study. J Clin Oncol 2025; 43(33): 3610-21 11 Varlotto JM et al.: EA5181: phase 3 trial of concurrent and consolidative durvalumab vs consolidation durva alone for unresectable stage 3 NSCLC. J Thorac Oncol 2025; 20(10): S4-5 12 Peters S et al.: CheckMate 73L: phase III study comparing nivolumab (N) + concurrent chemoradiotherapy (CCRT) followed by N ± ipilimumab (I) v CCRT followed by durvalumab (D) for previously untreated, locally advanced stage (stg) III NSCLC. Immuno-Oncology and Technology 2024; 24: 100808 13 Acker F et al.: Efficacy and safety of immune checkpoint inhibition combined with concurrent chemoradiotherapy in patients with stage III unresectable non-small cell lung cancer: a systematic review and meta-analysis. Eur J Cancer 2025; 218: 115266 14 Telarovic I et al.: Delayed tumor-draining lymph node irradiation preserves the efficacy of combined radiotherapy and immune checkpoint blockade in models of metastatic disease. Nat Commun 2024; 15(1): 5500 15 Dejonckheere CS et al.: Impact of estimated dose of radiation to immune cells (EDRIC) in locally advanced non-small-cell lung cancer: a secondary analysis of the multicenter randomized PET-Plan trial. Radiother Oncol 2025; 208: 110907 16 Aggarwal C et al.: Durvalumab alone or combined with novel agents for unresectable stage III non-small cell lung cancer: update from the COAST randomized clinical trial. JAMA Netw Open 2025; 8(7): e2518440 17 Provencio M et al.: Induction chemo-immunotherapy followed by chemo-radiotherapy and immunotherapy maintenance in stage III NSCLC (APOLO): a phase 2 trial. Nat Commun 2025; 16(1): 10124 18 Wang Y et al.: The preliminary results of a randomized phase II trial evaluating induction toripalimab plus chemotherapy followed by concurrent chemoradiotherapy and consolidation toripalimab in bulky unresectable stage III non-small-cell lung cancer (InTRist). J Clin Oncol 2025; 43(16_suppl): 8012 19 Qiu B et al.: Consolidative nivolumab versus observation in unresectable stage III non-small cell lung cancer patients following neoadjuvant nivolumab plus chemotherapy and concurrent chemoradiotherapy (CA209-7AL): a randomized clinical trial. Signal Transduct Target Ther 2025; 10(1): 317 20 Ross HJ et al.: Atezolizumab before and after chemoradiation for unresectable stage III non-small cell lung cancer: a phase II nonrandomized controlled trial. JAMA Oncol 2024; 10(9): 1212-9 21 Ohri N et al.: Selective personalized radioimmunotherapy for locally advanced non-small-cell lung cancer trial (SPRINT). J Clin Oncol 2024; 42(5): 562-70 22 Filippi AR et al.: Durvalumab after radiotherapy in patients with unresectable stage III non-small-cell lung cancer ineligible for chemotherapy: the DUART phase II nonrandomized controlled study. ESMO Open 2025; 10(9): 105560 23 Hata A et al.: Radiotherapy-free pembrolizumab combined with chemotherapy for locally advanced non-small-cell lung cancer with PD-L1 tumour proportion score of 50% or higher (Evolution trial): a multicentre, single-arm, phase 2 study. Lancet Oncol 2025; 26(11): 1432-42 24 Reck M et al.: Neoadjuvant durvalumab (D) + chemotherapy (CT) followed by either surgery (Sx) and adjuvant D or CRT and consolidation D in patients with resectable or borderline resectable stage IIB-IIIB NSCLC: interim analysis (IA) of the phase II MDT-BRIDGE study. Ann Oncol 2025; 36: 1606
Das könnte Sie auch interessieren:
Erhaltungstherapie mit Atezolizumab nach adjuvanter Chemotherapie
Die zusätzliche adjuvante Gabe von Atezolizumab nach kompletter Resektion und adjuvanter Chemotherapie führte in der IMpower010-Studie zu einem signifikant verlängerten krankheitsfreien ...
Highlights zu Lymphomen
Assoc.Prof. Dr. Thomas Melchardt, PhD zu diesjährigen Highlights des ASCO und EHA im Bereich der Lymphome, darunter die Ergebnisse der Studien SHINE und ECHELON-1
Aktualisierte Ergebnisse für Blinatumomab bei neu diagnostizierten Patienten
Die Ergebnisse der D-ALBA-Studie bestätigen die Chemotherapie-freie Induktions- und Konsolidierungsstrategie bei erwachsenen Patienten mit Ph+ ALL. Mit einer 3-jährigen ...
.jpg)